FDA approved Lupus drugs
Clinical trials are an incredibly important contribution to new drugs for lupus. Before 2011, there had not been any new FDA approved drugs for systemic lupus in over 50 years. Before that there were only three, aspirin, plaquenil and prednisone. Now, with Benlysta, there are four. New biologic drugs like Benlysta are constantly being researched and we will likely have more soon. We all hope it won’t take another 50 years to get number five! In the mean time, there are still things we can do to improve treatment.
One of the drawbacks to Benlysta is the great cost for the drug and the infusions. It is mixed following exacting rules before infusion, taking at least thirty minutes to prepare. Living within walking distance from my new rheumatologist, I am also within each reach of the research arm of the practice. The practice is participating in a handful of lupus related research trials, including some patients from the first Benlysta clinical trials. The next chapter of my lupus treatment will mean a break in the monthly infusion routine.
What comes next?
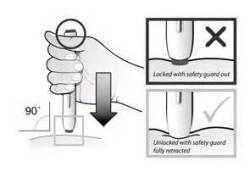
After having my last infusion a few days ago, in about ten more days weekly self-injections of 1/4 the amount of Benlysta will begin to replace monthly infusions. The next couple of months will be spent participating in a clinical trial to test an auto injector that delivers Benlysta as a subcutaneous injection. Last week, I had my training in how to use the auto injector and shot (wasted) a whole real dose of Benlysta into a fake “skin” device to prepare for my first self-injection. The plan is to do the first and two of the other eight injections under the watchful tutoring of the research nurse, and the rest of my weekly infusions are at home.
Official information about the clinical trial
GlaxoSmithKline’s Phase II Open-Label Single-Arm Study to Evaluate the Reliability of an Autoinjector that Administers Belimumab Subcutaneously in Subjects with Systemic Lupus Erythematosus (SLE)
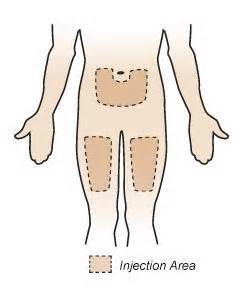
The primary outcome of the study is to assess the suitability of the autoinjector for self-administration of belimumab by subjects with SLE in real-life conditions. The study will assess the use of the autoinjector inside the clinic setting and outside the clinic setting. The study will also assess the safety and tolerability of belimumab administered subcutaneously (SC) via the autoinjector. Subjects will self-administer belimumab SC into the thigh or abdomen using the autoinjector device for 8 weekly doses. Subjects will return for a follow-up visit 4 weeks after the last SC dose of belimumab. All injections will be assessed by the investigators for success based on direct observation and/or the subject diary. A total of 118 subjects (treated with at least one dose of study drug) are planned to be enrolled in this study. The study will last through mid-2015 and is being done in these cities:
Gilbert and Phoenix, Arizona
Lansing, Michigan
Charlotte and Greenville, N. Carolina
Greenville, S. Carolina
Nearly foolproof auto injector
It seems like this will be nearly foolproof. I am no stranger to “Sub Q” injections, after giving scores of them the old-fashioned way helping my husband with blood thinning Lovenox injections. After a bed-side crash course in his hospital room a few years ago, I became a pro at using short-needles with pre-filled injections, and jabbing him in the tummy to help stave off the return of massive blood clots. I stayed pretty calm doing them, so I have no apprehension about doing them with an auto injector with Benlysta. In comparison, the new auto injector seems like child’s play. Just like many other “Sub Q” injections, it is injected somewhere either in a U-shaped area around the navel, or in either upper thigh.
Fewer flares?
Because with the monthly infusions I tend to flare during the last week, it will be great to find out if evening out the dose to weekly injections will help prevent the recurring monthly flare cycle. I will be keeping a journal as part of the study, so look for updates here, too.
It is my hope that by my participation I will help decrease the costs of getting Benlysta to the people who need it most. One bonus? During my participation in the clinical trial, there is no cost for seeing my rheumatologist, lab work or the medication. Also, at the end of the trial, there will be a small stipend coming to me of $5o for each in-office visit during the study. There is always room in my wallet for a little fun money!













Comments on: "Benlysta for Lupus Without Infusions?" (14)
I can’t get any help because I’m on Medicare
Income over
Melissa, Thank you so much for responding to this post. The issues about coverage and Medicare concern me, too, as I am approaching retirement. Right now, just grateful for the insurance benefits that go with my job. LA
Are the trials still going?
Is there a way I can participate?
When the Benlysta injection come out?
I just got my last clinical trial Benlysta infusion today. My doctor wants to put me on the injections but they don’t exist yet. I don’t want to go into a flare & have to go on prednisone. Prednisone has not been my friend where I might be needing knee replacement in the near future.
Thank you in advance for any information you can give.
Sincerely,
Tomaka
According to Dr Donald Thomas, MD, author of “The Lupus Encyclopedia “, the Benlysta shots (you can self inject) are as effective as the infusions and he highly recommends them. I’ll get a link —
How many injections at a time? One or two? Is this just one shot a week?
There was one injection each week. LA
This is exciting news!! The last week before the infusion is very painful for my daughter. Weekly shots I hope would really help with that. God Bless You!!!! Thank you for this information and your updates on your progress.
Is the self injections out yet
K, I am not sure, although I read a recent medical journal article this week that said the results from injections seemed to be equally effective as the monthly infusions. That would be a great research topic for an upcoming post… thank you so much for your comment and question. We will see what we can do to get a good answer. LA
Hi, I was wondering how the trial is going. 🙂 I read an article that said the first set of results was to be released towards the end of 2014. ? Hope it’s been great and that we can look forward to it soon!
Eve, I am not sure how the trial has come out, but that is a great question. I will see what I can find out. Thanks for your comment and joining the conversation! LA
The million dollar question is how will insurance pay for this? Will it be considered a prescription whereas I have a co payment? Or how it is now, where the in office infusion I have no co payment. If it is considered a precription like from a drug store, this treatment may become unaffordable for many.
I am looking forward to hearing how this goes. How great you could participate in this study. I hope it goes well for you. Thanks for all the great info.
DYD, Thanks! I had my second injection this week, and it went well. So far, I feel better than I would normally. This would have been the start of my 4th week between infusions, and I don’t feel the flare coming on that I usually have. So far, so good. I did inject on the border of an old deep scar and it hurt more than I expected, but that passed very quickly. The first week, I could hardly feel it. No bad reactions at all. Woo hoo! LA